
Throughout most of his life, Thomson was a leading scientific figure in Britain. Research since that time has resulted in the abandonment of the Rutherford model in favor of other atomic models. The Rutherford atomic model described the structure of the atom as a positively charged nucleus around which negatively charged electrons circulated. He maintained this notion until experimental research and theoretical work indicated that the atomic model described in 1911 by Ernest Rutherford, a former student of Thomson, was much more likely. According to Thomson’s view, each atom was a positively charged sphere with electrons scattered throughout (like bits of fruit in a plum pudding). Thomson was initially a strong proponent of what is commonly called the plum-pudding atomic model or the Thomson atomic model, although many other representations of the atom were suggested by his contemporaries. Once the existence of the electron was accepted, the next step was to consider how the particles were incorporated into the atom. These and additional studies carried out by Thomson and others quickly led to widespread acceptance of Thomson’s discovery. In one experiment, Thomson attempted to use magnetism to see if negative charge could be segregated from cathode rays, in another he tried to deflect the rays with an electric field, and in a third he assessed the charge-to-mass ratio of the rays. The evidence he produced in support of his theoretical claims was culled from a series of innovative experiments with cathode ray tubes. On April 30, 1897, Thomson made his discovery public while giving a lecture to the Royal Institution. That same year, when Thomson returned to Cambridge, he made his most significant scientific discovery, that of the electron (which he initially referred to as the corpuscle). In 1897, the lectures were published as Discharge of Electricity through Gases. 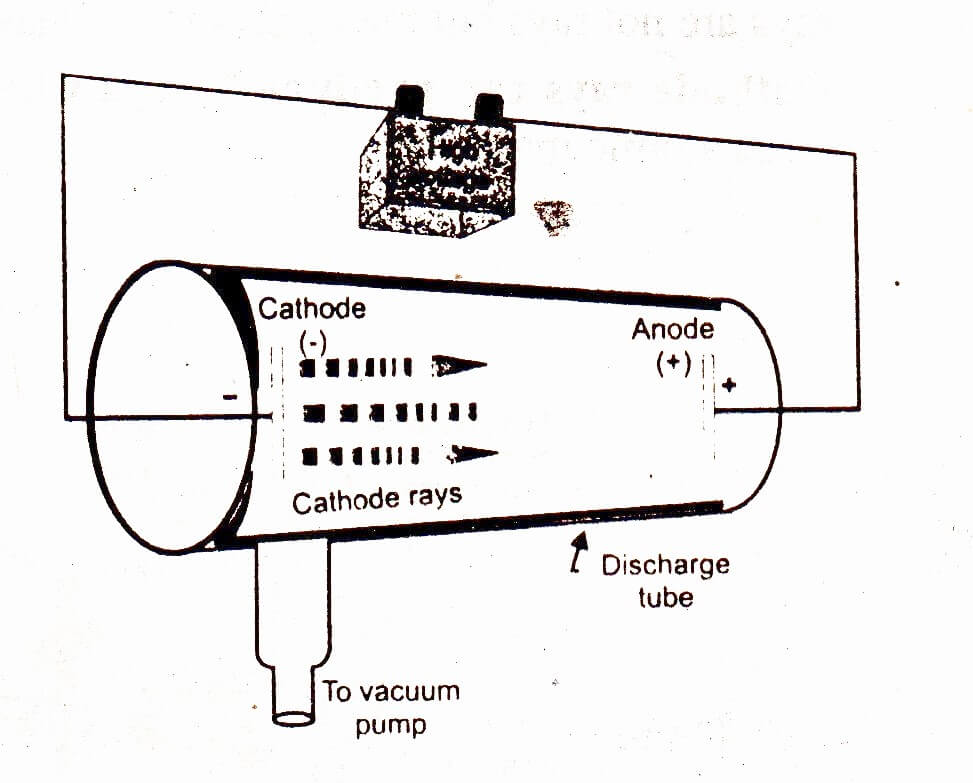 
During a visit to the United States in 1896, he gave a series of lectures discussing his findings. In the early 1890s, much of Thomson’s research focused on electrical conduction in gases. Coverage of these efforts, which continued over many years, appeared in Thomson’s 1892 treatise Notes on Recent Researches in Electricity and Magnetism. The receipt of such considerable honors by so young a scientist was highly unusual, but was largely the result of Thomson’s significant early work expanding James Clerk Maxwell’s theories of electromagnetism. Also in 1884, the Royal Society of London elected Thomson as a Fellow. In 1883, he became a lecturer at Cambridge and the following year was appointed Cavendish Professor of Experimental Physics, becoming the successor to Lord Rayleigh. In 1880, Thomson received a bachelor’s degree in mathematics and became second wrangler, a title bestowed on the second highest-scoring individual on the Cambridge mathematics exams.įollowing graduation, Thomson became a Fellow at Trinity College and began work at the Cavendish Laboratory, part of the Cambridge Physics Department. He remained associated with Cambridge University in varying capacities the rest of his life. He then transferred to Trinity College, Cambridge, on a mathematics scholarship. However, through the efforts of his family and scholarships he continued at Owens College until 1876. Thomson’s father died only a few years into his college studies, making it financially difficult for Thomson to remain in school. He entered Owens College when he was 14 years old, where he became interested in experimental physics, though he had initially intended to pursue a career in engineering. The son of a bookseller, Thomson was born on December 18, 1856, in Cheetham Hill, located just north of Manchester, England. Thomson received the Nobel Prize in Physics in 1906 for his work exploring the electrical conductivity of various gases. A series of experiments with cathode rays he carried out near the end of the 19th century led to his discovery of the electron, a negatively charged atomic particle with very little mass. Thomson, was a British physicist who first theorized and offered experimental evidence that the atom is a divisible entity rather than the basic unit of matter, as was widely believed at the time.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
